Can Novel Coronavirus Be Cure or Treated: China has launched a clinical preliminary to expediently test medication for the novel coronavirus disease as the country surges treatments for those distressed and scours for antibodies to ensure the rest.
Remdesivir, another antiviral medication by Gilead Sciences Inc. focused on irresistible maladies such as Ebola and SARS, will be tried by a medicinal group from Beijing-based China-Japan Friendship Hospital for adequacy in treating the lethal new strain of coronavirus, a clinic representative revealed to Bloomberg News Monday.
Preliminary for the medication will be led in the focal Chinese city of Wuhan – ground zero of the viral episode that has so far slaughtered in excess of 360 individuals, sickened more than 17,000 in China and spread to in excess of twelve countries. Upwards of 270 patients with gentle and moderate pneumonia brought about by the infection will be enrolled in a randomized, twofold blinded and fake treatment controlled examination, Chinese news outlet The Paper provided details regarding Sunday.

Gilead shares rose as much as 8.7% in New York, their greatest intraday gain since November 2012. A year ago, the stock wrapped up 3.9%, failing to meet expectations of the 24% addition in the Nasdaq Biotechnology Index.
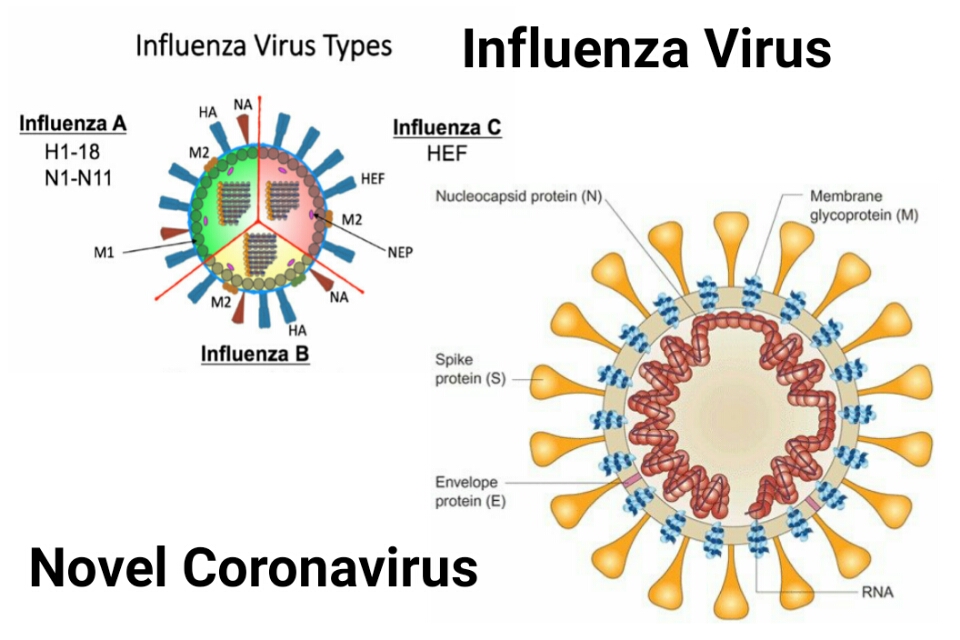
Difference between Influenza virus and Novel Coronavirus
While financial specialists are bullish, it commonly takes a very long time to explore and build up a novel drug for any sickness, with viral diseases presenting specific difficulties. The primary test drugs utilized against Ebola, including redeliver and Mapp Biopharmaceutical Inc’s. ZMapp, in the end, yielded disillusioning outcomes.
There are still no medicines affirmed for those contaminated with Ebola, over five years after they were first given to patients on a humane premise since nothing else was accessible.
Desires for the medication have been floated by reports that the main patient in the U.S. contaminated with the infection, a 35-year-old Washington state man, saw his pneumonia improve after he was given redeliver, specialists treating him said in an investigation distributed in the New England Journal of Medicine a week ago.
However, it’s imaginable too early to give the medication kudos for having any kind of effect all things considered, as indicated by examiners at Raymond James. The infection had just begun to subside before the patient was given the medicine, they wrote in a note to customers Monday.
Drugmakers, for example, GlaxoSmithKline Plc. just as Chinese specialists are dashing to crash create immunizations and treatments to battle the new infection that is more infectious than SARS and could cost the worldwide economy multiple times more than the $40 billion sapped by the 2003 SARS flare-up. The choice to hold human preliminaries for remdesivir shows it’s among the most encouraging treatments against the infection that so far has no particular medications or antibodies.
“Gilead is by all accounts now among the leaders in building up treatment for coronavirus,” Mohit Bansal, an investigator with CitiGroup Inc., said in a note to customers. He said it could take a long time for any item to arrive at the market, however, that “we could see a business open door for Gilead in longer-term too since normally nations store such medications to shield against future flare-ups.”
Can Novel Coronavirus Be Cure or Treated
Avoidance, Treatment of Novel Coronavirus (2019-nCoV) If you are looking for how to cure novel coronavirus then there is currently (Feb 3, 2020) no vaccine available to prevent 2019-nCoV disease. The most ideal approach to prevent contamination is to avoid from being presented or close contact with this infection or an infected person. Nonetheless, as an update, the CDC consistently prescribes ordinary preventive activities to help forestall the spread of respiratory infections, including:
- Wash your hands regularly with cleanser and water for at any rate 20 seconds, particularly subsequent to heading off to the restroom; before eating; and in the wake of cleaning out your nose, hacking, or sniffling.
- On the off chance that cleanser and water are not promptly accessible, utilize a liquor based hand sanitizer with at any rate 60% liquor. Continuously wash hands with cleanser and water if hands are unmistakably filthy.
- Avoid contacting your nose, eyes, and mouth.
- Maintain a distance from contact with the person who is wiped out or with flu.
- Remain at home when you are with flu or wiped out.
- cover your sneeze and cough with tissue to avoid the spreading of this virus.
- Clean and sanitize every now and again contacted articles and surfaces utilizing a normal family unit cleaning shower or wipe.
Exploratory Drug
The exploratory medication has not yet been affirmed for use by any medication controller on the planet yet is being utilized on patients fighting the new infection without endorsed treatment choices, Gilead said in an announcement a week ago. Gilead is working with wellbeing experts in China to set up the preliminary to decide if remdesivir can securely and adequately be utilized for this contamination, it said.
China’s wellbeing controller has likewise prescribed AbbVie Inc’s HIV medication Kaletra as a specially appointed antiviral medication for coronavirus. Kaletra is likewise set to experience human preliminaries, as indicated by The Paper.
Then, a worldwide quest proceeds for treatments to contain the disease that can spread undetected.
Johnson and Johnson have started chip away at a preventive coronavirus antibody and has “many researchers” dealing with it, it’s Chief Scientific Officer Paul Stoffels said a month ago. GlaxoSmithKline and the Coalition for Epidemic Preparedness Innovations said Monday they will work to quicken the production of an antibody and afterward give the portions quickly.
The Coalition, set up in 2017 to spike the improvement of shots for known ailments and to react to new infections, has likewise marked agreements with drugmakers including Moderna Inc. what’s more, Inovio Pharmaceuticals Inc. as ahead of schedule as of Jan. 22 to facilitate a deal with antibodies. Novavax Inc. was among the initial ones to report it was taking a shot at an applicant as well.
Forefront Treatment
Wellbeing authorities, in any case, say an immunization variant may take three months to be accessible for the primary phases of human testing while at the same time building up a compelling antibody for the most part takes years.
That puts remdesivir on the bleeding edges of fighting the disease.
(Can Novel Coronavirus Be Cure or Treated) – The preliminary in China could prompt a most optimized plan of attack endorsement of remdesivir by the Chinese medication controller, which at times has been the quickest on the planet. China’s medication law currently permits contingent endorsement for drugs with clinical information exhibiting viability against aliments such are reality compromising and have no current treatments.
source